Articles by Shari Barbanel
Industry Responds to Oregon AG Lawsuit Against GNC
The natural products industry is responding to a lawsuit filed by Oregon Attorney General (AG) Ellen Rosenblum alleging that supplement retailer GNC (Pittsburgh, PA) violated the state’s Unlawful Trade Practices Act. The lawsuit accuses GNC of selling workout supplements containing picamilon, a prescription drug used to treat neurological conditions. Acting Deputy Director of the Dietary ...
Kerry Acquires Wellmune
Kerry Group plc (Tralee, Ireland) has acquired Wellmune from Biothera Inc. (Eagan, MN), a natural food, beverage and supplement ingredient that is clinically proven to help strengthen the immune system. “At Kerry, cutting-edge science and technology informs everything we help shape and create. The addition of Wellmune to our portfolio offers Kerry tremendous growth opportunities ...
Deerland Enzymes Joins NASC
Deerland Enzymes (Kennesaw, GA) has announced the company’s acceptance into membership of the National Animal Supplement Council (NASC) as a Preferred Supplier. Founded in 2001, NASC is a non-profit trade organization dedicated to protecting and enhancing the health of companion animals and horses throughout the United States. Deerland Enzymes joins a membership that includes manufacturers ...
DDW Opens New Natural Food Color Operation
DDW (Louisville, KY), “The Color House,” has announced the opening of its second site in Louisville for manufacturing food ingredients. Located next to the company’s global support center on Spring Street and near its caramel color operation and U.S. subsidiary on Payne Street, the facility will produce specialty colorings for leading food and beverage companies ...
PLT Health Solutions Launches Transparency & Trust-building Initiative
PLT Health Solutions, Inc. (Morristown, NJ) announced it has launched a formal transparency and trust-building initiative for its ingredient portfolio. The initiative is designed to support emerging opportunities for food, beverage and supplement companies that can deliver assured ingredient quality, demonstrable efficacy, and clean labels in consumer products. Called PLT360, the initiative has a major ...
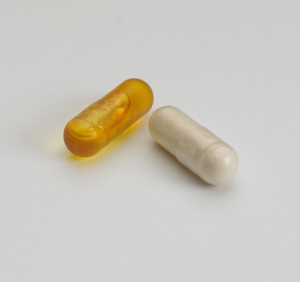
Capsugel Expands DRcaps Capsule Platform
Capsugel (Morristown, NJ) has announced that it has developed a technology that makes its vegetarian, acid-resistant DRcaps hard capsules suitable for liquid-fill applications. As a result, Capsugel’s customers can now bring the benefits of DRcaps capsules—including resistance to stomach acid and taste/odor-masking—to liquid-based dietary supplements. This new offering will be featured at SupplySide West 2015 ...
Sabinsa Honored With Two Pharmexcil Awards
Sami Labs (India), the manufacturing arm of Sabinsa Corporation (East Windsor, NJ), has received two Pharmexcil Awards for 2014-2015 in the Herbals category: the Silver Award for Patents and the Outstanding Export Performance Award. Pharmexcil, a ministry set up by the Government of India as a gateway to global pharma trade, bestows annual awards to ...
NSF Launches Suite of GMO Transparency Services
Responding to industry and consumer demand for non-GMO (genetically modified organism) certification options, NSF International has launched NSF Non-GMO True North. NSF International’s newest certification offering expands its suite of GMO transparency services for retailers, manufacturers, suppliers and producers. The new non-GMO certification, available through NSF International’s Consumer Values Verified Program, is offered in addition to ...
NYAG Targets Devil’s Claw Supplements, Industry Responds
On September 9, New York Attorney General (NYAG) Eric Schneiderman sent cease-and-desist letters to 13 companies that sell devil’s claw herbal dietary supplements informing them that their products containing devil’s claw material were tested at the New York Botanical Garden using DNA technology. The results showed that the devil’s claw was a different botanical species ...
ADM Names New Leaders of Wild Flavors, Specialty Commodities Businesses
Archer Daniels Midland Company (ADM, Chicago, IL) recently named Kenneth Campbell president of the company’s WILD Flavors business, and named Manda Tweten president of the company’s Specialty Commodities business (SCI). Campbell, who most recently served as president of ADM’s Specialty Commodities business, brings 18 years of experience with ADM to his new role, having previously ...
Don't Miss Out!
Industry Professionals
Stay Informed!
Stay informed about the latest health, nutrition, and wellness developments by signing up for a FREE subscription to Nutrition Industry Executive magazine and digital newsletter.
Once subscribed, you will receive industry insights, product trends, and important news directly to your doorstep and inbox.