fda
FDA Commissioner Dr. Marty Makary Resigns
On May 12, U.S. Food and Drug Administration (FDA) Commissioner Dr. Marty Makary resigned from his position after 13 months. As reported by The New York Times, he faced mounting pressure and criticism, ultimately leaving over the Trump administration’s plans to authorize fruit-flavored e-cigarettes and vapes. Makary is a surgical oncologist, professor, researcher, author and medical ...
Industry Responds to Introduction of New Dietary Supplement Listing Act
On April 20, U.S. Representative Maxine Dexter, MD (D-OR) introduced a new Dietary Supplement Listing Act to require dietary supplement manufacturers to register their products with the U.S. Food and Drug Administration (FDA). A similar bill was introduced in 2024 and then reintroduced in 2026 by Senator Dick Durbin (D-IL). The Dietary Supplement Listing Act ...
FDA Holds Public Meeting Exploring Scope of Dietary Supplement Ingredients
On March 27, the U.S. Food and Drug Administration (FDA) hosted a public meeting on the scope of dietary supplement ingredients. The event was held in-person from 9 a.m. to 3 p.m. EST at Wiley Auditorium, 5001 Campus Dr. in College Park, MD and virtually. FDA’s Office of Dietary Supplement Programs (ODSP) invited stakeholders to participate in ...
U.S. Representative Introduces Dietary Supplement Regulatory Uniformity Act
On Feb. 4, U.S. Representative Nick Langworthy (R-NY) introduced the Dietary Supplement Regulatory Uniformity Act, legislation aimed at protecting consumer health while restoring common sense to dietary supplement regulation by reaffirming a single, science-based national standard overseen by the U.S. Food and Drug Administration (FDA). According to Langworthy, dietary supplements including vitamins, protein powders, creatine ...
Nammex Renews Call for Functional Fungi Label Accuracy and Transparency in Response to ‘Misinformation Campaign’
In what it calls a “misleading marketing campaign,” Nammex (Gibsons, BC, Canada) said it feels compelled to renew a call for accuracy and transparency in labeling and marketing fungi products. “As happens more often than it should, those of us who have long been advocates for transparency in fungi product labeling and marketing are compelled ...
Dick Durbin Reintroduces Dietary Supplement Listing Act
On Jan. 15, U.S. Senator Dick Durbin (D-IL) reintroduced the Dietary Supplement Listing Act, which would require dietary supplement manufacturers to register their products with the U.S. Food and Drug Administration (FDA). According to Durbin, the law would require companies to provide FDA with critical information about their products including product names, a list of ...
FDA Issues Letter on DSHEA Disclaimer Amendment, Industry Responds
On Dec. 11, the U.S. Food and Drug Administration (FDA) published a letter to the dietary supplement industry regarding the agency’s labeling regulations that govern the placement of the disclaimer on dietary supplements labels under the Federal Food, Drug and Cosmetic Act (FD&C) and Dietary Supplement Health and Education Act of 1994 (DSHEA). The agency ...
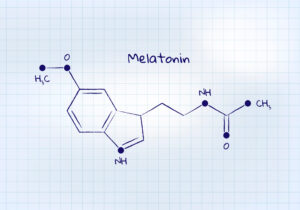
Associations Respond to Melatonin Study Presented at the 2025 AHA Scientific Sessions
Long-term use of melatonin supplements, often used to promote sleep and address insomnia, was associated with a higher risk of heart failure diagnosis, heart failure hospitalization and death from any cause in chronic insomnia, according to a preliminary study to be presented at the American Heart Association’s (AHA) Scientific Sessions 2025. In the study, researchers classified people who ...
Anresco Laboratories and NJ Labs Unite to Form Nationwide Testing Network
Anresco Laboratories (San Francisco, CA) and NJ Labs (New Brunswick, NJ) have joined forces to establish Anresco NJ Labs, a nationwide network uniting trusted, compliant and independent testing solutions across multiple regulated industries. Combining nearly two centuries of proven expertise, Anresco NJ Labs brings together the comprehensive scientific experience of Anresco Laboratories, founded in 1943 and ...
FDA Declares NMN Lawful in Dietary Supplements, Industry Reacts
The U.S. Food and Drug Administration (FDA) has reversed course and confirmed that Beta (β) Nicotinamide Mononucleotide (NMN) is lawful for use in dietary supplements. Responding to citizen petitions filed by the Natural Products Association (NPA), Council for Responsible Nutrition (CRN), and the Alliance for Natural Health (ANH), the FDA revised its interpretation of a ...
Don't Miss Out!
Industry Professionals
Stay Informed!
Stay informed about the latest health, nutrition, and wellness developments by signing up for a FREE subscription to Nutrition Industry Executive magazine and digital newsletter.
Once subscribed, you will receive industry insights, product trends, and important news directly to your doorstep and inbox.