Natural Products Association
FDA Declares NMN Lawful in Dietary Supplements, Industry Reacts
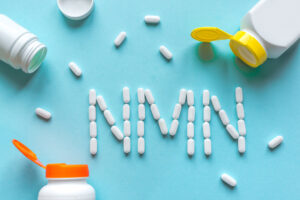
The U.S. Food and Drug Administration (FDA) has reversed course and confirmed that Beta (β) Nicotinamide Mononucleotide (NMN) is lawful for use in dietary supplements. Responding to citizen petitions filed by the Natural Products Association (NPA), Council for Responsible Nutrition (CRN), and the Alliance for Natural Health (ANH), the FDA revised its interpretation of a ...
NPA Launches Revamped Website
The Natural Products Association (NPA) has modernized its website. Designed as a cornerstone of NPA’s multimedia strategy, the modernization of the NPA website delivers relevant, timely content for members, the broader natural products industry, and other stakeholders as the association approaches its 90th-year anniversary in 2026. What’s New: 90 Years of Natural: A dedicated section ...
Trade Associations Send Joint Letter Regarding FSA/HSA Proposals
The American Herbal Products Association (AHPA), the Council for Responsible Nutrition (CRN), the Consumer Healthcare Products Association (CHPA) and the United Natural Products Alliance (UNPA) sent a joint letter to the Internal Revenue Service (IRS) to obtain coverage for dietary supplements under Flexible Spending Accounts (FSAs) and Healthcare Savings Accounts (HSAs). There is also a ...
NPA Urges IRS to Include Supplements as Qualified Medical Expenses
The Natural Products Association (NPA) urged the Internal Revenue Service (IRS) to modernize IRS Publication 502 to expand “qualified medical expenses” to include nutritional and dietary supplements that carry U.S. Food and Drug Administration (FDA)-authorized health claims or structure-function claims. This update would allow consumers to use their Health Savings Accounts (HSAs), Flexible Spending Accounts ...
FDA Issues Warning Letters to Firms Marketing Products Containing 7-Hydroxymitragynine
On July 15, the U.S. Food and Drug Administration (FDA) announced seven recent warning letters sent to companies for illegally marketing products containing 7-hydroxymitragynine, also known as 7-OH. This action reflects the Agency’s growing concern around novel potent opioid products being marketed to U.S. consumers and sold online and in smoke shops, gas stations and ...
NPA Responds to Taurine Study
In a new study, published in the journal Nature, researchers from the University of Rochester Medical Center said that they identified taurine, which is made naturally in the body and consumed through some foods, as a key regulator of myeloid cancers such as leukemia. The Wilmot Cancer Institute investigators at the University of Rochester were ...
Natural Products Industry Responds to Trump Tariffs
On April 2, President Donald Trump declared that “foreign trade and economic practices have created a national emergency.” His executive order imposes responsive tariffs to “strengthen the international economic position of the United States and protect American workers.” Under the order, Trump imposed a 10 percent tariff on all countries effective April 5. There will ...
White House Announces Tariff Exemptions for Key Dietary Supplement Ingredients
The Trump administration has announced that certain dietary supplement and dietary ingredients have been excluded from the Harmonized Tariff Schedule (HTS) announcement on April 2. In the executive order, President Trump provided a list of goods and materials (Annex II) that are classified in the 8-digit subheadings of the HTS that are exempt. Some of ...
NOW Health Group’s Jim Emme Appointed Chairman of the NPA Board of Directors
The Natural Products Association (NPA) has announced that NOW Health Group (Illinois) CEO Jim Emme has been appointed as the new Chairman of the NPA Board of Directors. This appointment marks a new chapter in the longstanding partnership between NOW Health Group and NPA, furthering their commitment to advancing the natural products industry through advocacy, ...
Trade Associations Congratulate RFK Jr. on HHS Secretary Confirmation
On Feb. 13, politician and environmental attorney Robert F. Kennedy Jr. (RFK) was confirmed as the next Secretary of Health and Human Services (HHS). The Natural Products Association (NPA), the Council for Responsible Nutrition (CRN), the American Herbal Products Association (AHPA) and the Consumer Healthcare Products Association (CHPA) congratulated Kennedy on his confirmation and expressed ...
Don't Miss Out!
Industry Professionals
Stay Informed!
Stay informed about the latest health, nutrition, and wellness developments by signing up for a FREE subscription to Nutrition Industry Executive magazine and digital newsletter.
Once subscribed, you will receive industry insights, product trends, and important news directly to your doorstep and inbox.